Keynote Summaries
On GERM 3, La Jolla, March 6-9, 2001
Global Chemical Fluxes in Rivers
Speaker : Jerome Gaillardet
Student Writer : Graham Shields
Introduction
Presented very much as a group effort, Jerome Gaillardet’s (IPG Paris) oral contribution covered potamochemistry from 1) river discharge into the oceans, with boron as a case study, to 2) silicate weathering and its influence on the carbon cycle. In addition, some implications were discussed for the composition of the continental crust. Based on chemical analyses of both major and minor rivers worldwide, Gaillardet et al. outlined how river chemistry can be used to constrain the effects of carbonate versus silicate weathering on, for example, CO2 consumption, while emphasizing that refined riverine budgets are an essential first step to understanding ocean chemistry.
River Discharge
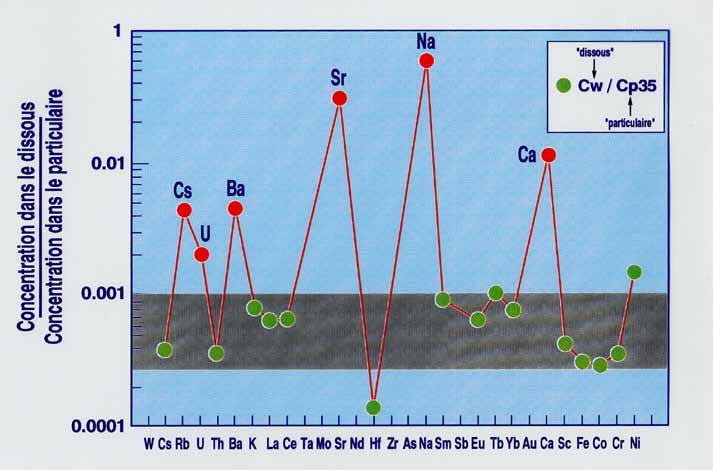
By analyzing elemental concentrations in river water, we can estimate the flux of these elements into the ocean. However, such calculations must overcome problems caused by the highly variable partitioning between the particulate and the dissolved fractions, which for many elements depends on filter size. This dependence is related to the existence of an organic colloidal phase that complexes the most insoluble elements and enhances their apparent solubility in natural waters. Some elements, such as Na, Sr, Ca, Rb, Ba, U and B do not generally change concentration with filter size (Figure 1), implying that they are consistently soluble, not affected by organic complexation and can thus be used to estimate river discharge. Preferential partitioning of these elements into the dissolved load results in their permanent depletion in the particulate load (Figure 5)
Boron is highly soluble, and so the riverine flux of B to seawater can be more easily estimated than for most elements. By analyzing most of the major rivers in the world, Lemarchand et al. (2000, Nature, 408, 951-954) were able to calculate that the input of B to the ocean is dominated by rivers, reaching approximately 38 x1010 g/yr compared with 6 x1010 g/yr for all other sources combined. Removal of B from the ocean is carried out by the weathering of ocean crust, adsorption on suspended particles, and carbonate deposition.
The d11B composition of riverine input lies close to +10‰, with an overall range between 0‰ and 22‰, seawater d11B = +40‰, while continental crust averages -10‰. The authors use a global ocean model, which takes into account the very long residence time of B in seawater (about 20 Ma), to estimate how seawater d11B is likely to have changed over the past 100 Ma. The resulting trend (Figure 2) matches published 11B data from foraminifera that cover the last 60 Ma (Pearson and Palmer, 1999, Science, 284,1824-1826), indicating that secular trends in seawater d11B may be used as a global weathering proxy.
One complication when modeling the B system is that measured d11B is also dependent on the partitioning between charged and uncharged borate species, which itself depends on pH. Therefore, trends in measured d11B in calcite, which are assumed to record solely the d11B of the ion B(OH)4-, may relate to changing seawater pH rather than B fluxes. During the discussion, the point was made that d11Bshifts, considerably more rapid than the c. 20 Ma oceanic residence time of B. Also d11Bdifferences between contemporary samples, can be more confidently interpreted as pH-related, while longer term changes are more ambiguous (S. Goldstein, T. Elliot). In addition, the leap of faith, which is required between interpreting d11Btrends as pH changes, and relating these pH changes to pCO2 could well turn out to be unjustified (L. Derry).
Silicate Weathering
Conventional wisdom dictates that climate change is strongly linked to the concentration of “greenhouse” gases in the atmosphere. The atmospheric budget of CO2, which is one of the most important of these greenhouse gases, is controlled largely by the chemical weathering of silicate rocks.
Existing proxies of chemical weathering rates, such as 87Sr/86Sr, are no longer considered reliable due to the frequently dominant effect of lithology on river water composition. Figure 3 shows how solutes in river water are derived from different sources, which allows the ratio Ca/Na to be used to estimate the relative importance of silicate versus carbonate weathering within a catchment area. A linear relationship between Ca/Na and HCO3¯/Na allows us to calculate the relative contribution of silicate versus carbonate weathering to the concentration of bicarbonate ions in river water, and hence to CO2 drawdown.
Chemical weathering rates, thus calculated, show only modest correlation with temperature (Figure 4). This is possibly due to the existence of hot catchment areas in regions of limited denudation, e.g. W. Africa (Gaillardet et al.,1999, Chem. Geol., 159, 3-30). However, good correlation exists between chemical weathering, runoff and physical erosion rates (Figure 4). This relationship indicates that chemical weathering is strongly lithology dependent, with basaltic rocks, yielding by far the highest chemical weathering rates. This means that basaltic rocks have a disproportionately large effect on CO2 drawdown, being responsible for as much as 25% of the global carbon flux.. The discussion brought out also that basalt weathering, for the same reasons, may have a similarly disproportionate effect on seawater 87Sr/86Sr. This reverses previous assumptions that seawater 87Sr/86Sr necessarily increases as a response to increased chemical weathering (L. Derry). In addition, basalt weathering on submarine volcanic glass, which is also particularly rapid, remains relatively unconstrained (H. Staudigel). The more general point was also made that by overemphasizing the role of CO2 in climate change, we may be neglecting other greenhouse gases, such as H2O (J. Veizer).
Composition of Upper Continental Crust
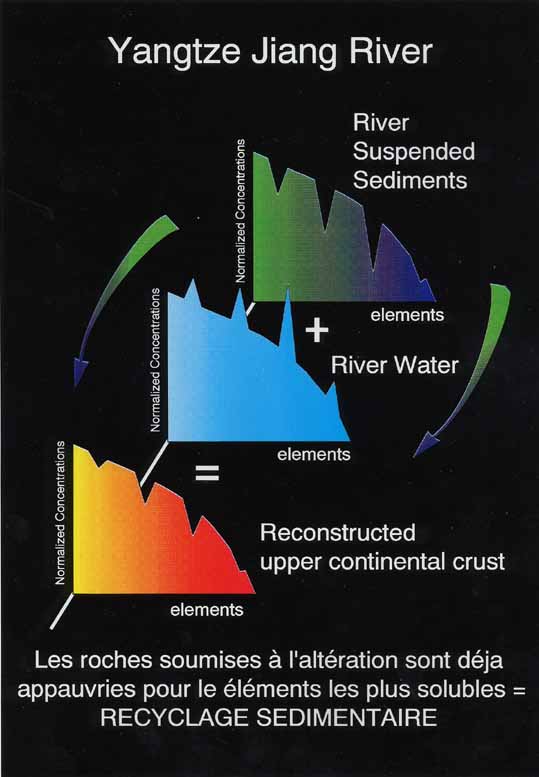
The mass budget that can be made for the most soluble elements between the dissolved and particulate phases for the largest rivers clearly shows that the continental crust which is subjected to chemical weathering does not have the conventional “granitic” composition. An important sedimentary reservoir (that has undergone previous weathering episodes) is necessary to account for the calculated chemical composition of the upper crust. Provided that estimates of the riverine physical denudation rates are known, the proportion of the recycled component can be estimated. As considerable amounts of sediment are deposited before entering the open marine realm on flood plains or in delta and estuarine areas, these are likely to be only minimum estimates (Gaillardet et al., 1999, G.C.A., 63, 4037-4051).
Conclusion
Further work is needed to 1) gather more data, 2) apply new isotopic tracers, and 3) constrain remaining uncertainties, such as temporal variability, solid river-borne flux, riverine organic chemistry, physical denudation rates, estuarine processes (the role of colloids) and reverse weathering in general.
Figures and Tables
Figure 1. Elemental partitioning between the dissolved and the particulate fraction in the River Congo.

Figure 2. The boron cycle (above) can be modeled, given that the temporal evolution of oceanic crust production, mechanical erosion and precipitation of marine carbonates are reasonably well known. The result (thick line below) matches measured δ11B data from foraminifera (Pearson and Palmer, 1999, Science, 284,1824-1826).
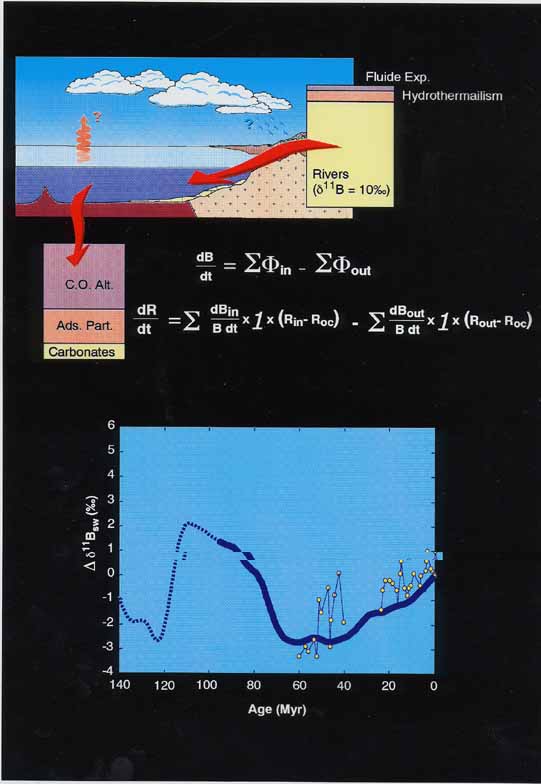
Figure 3. Origin of solutes in global mean river water.
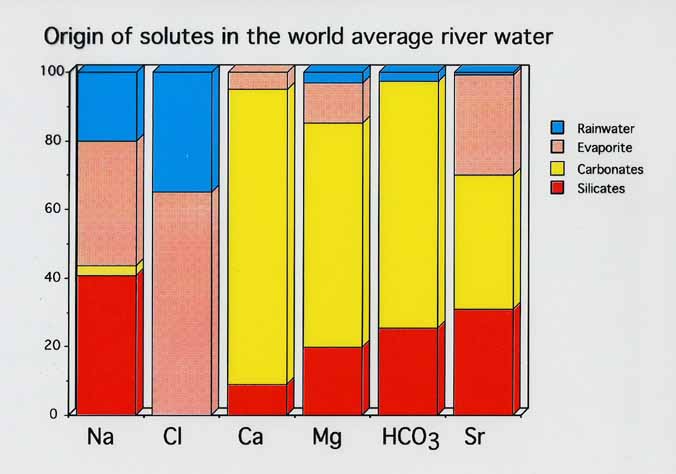
Figure 4. Chemical weathering rates shown against temperature and physical erosion rates.
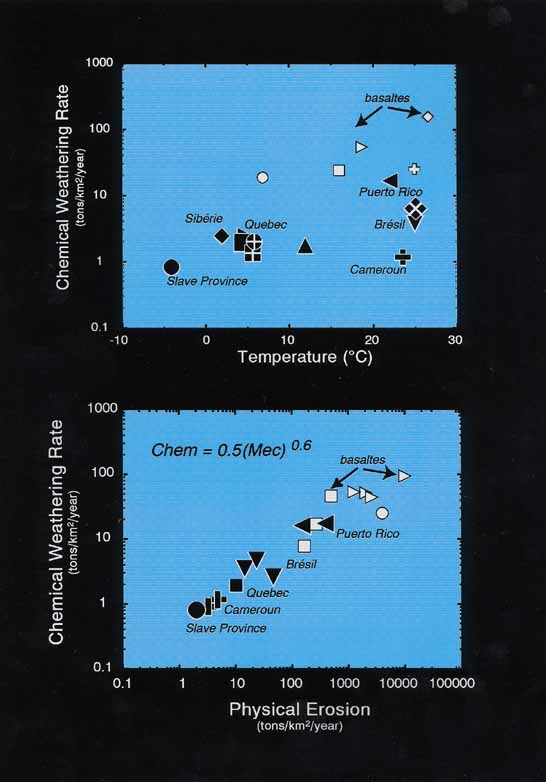
Figure 5. Depletion of soluble elements in river waters leads to their depletion in the suspended load.
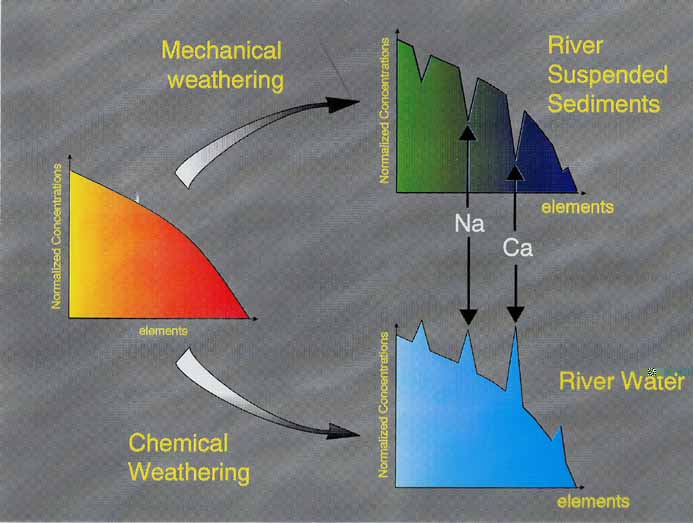
Figure 6. Sedimentary recycling of particulate matter depleted in soluble elements must lead eventually to their depletion in the upper continental crust.